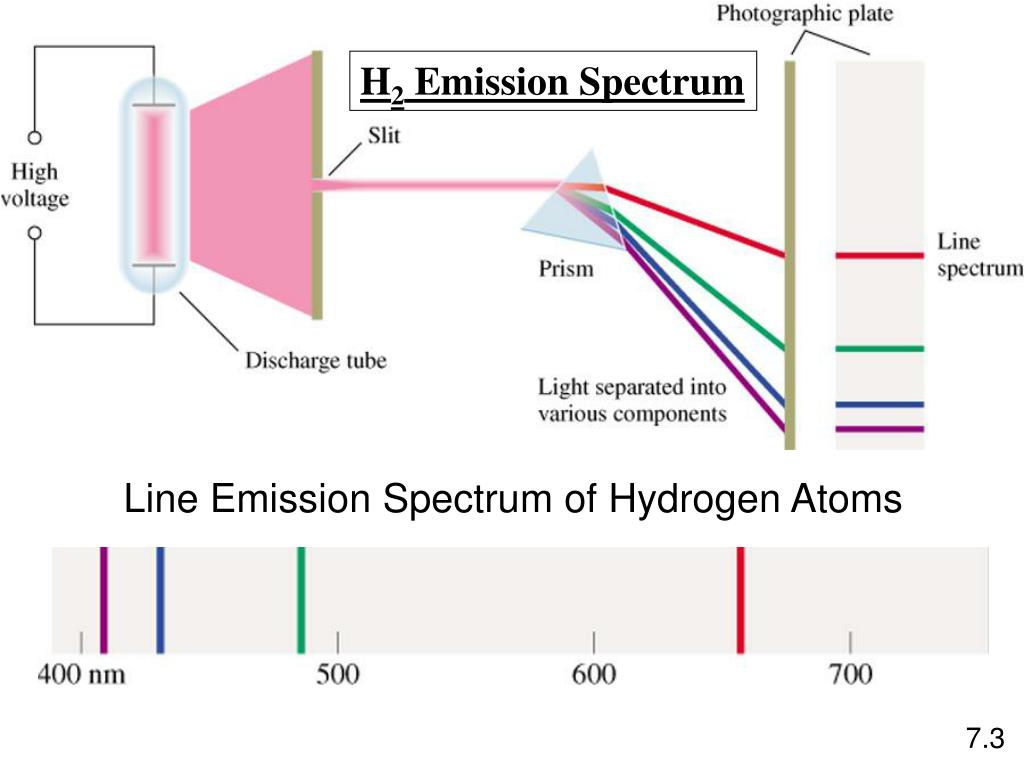
Line emission spectrum of hydrogen11/2/2023  The energy of an emitted photon corresponds to the energy difference between the two states. To distinguish the two states, the lower energy state is commonly designated as n′, and the higher energy state is designated as n. Spectral emission occurs when an electron transitions, or jumps, from a higher energy state to a lower energy state. The Bohr model was later replaced by quantum mechanics in which the electron occupies an atomic orbital rather than an orbit, but the allowed energy levels of the hydrogen atom remained the same as in the earlier theory. Each energy level, or electron shell, or orbit, is designated by an integer, n as shown in the figure. These states were visualized by the Bohr model of the hydrogen atom as being distinct orbits around the nucleus. 
The electromagnetic force between the electron and the nuclear proton leads to a set of quantum states for the electron, each with its own energy. Energy levels are not to scale.Ī hydrogen atom consists of an electron orbiting its nucleus. Bohr's model and postulates do not explain this phenomenon.Further information: Hydrogen atom Electron transitions and their resulting wavelengths for hydrogen.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
